
Treatment of preterm infants in whom the blood vessel that supplies oxygen to the body before birth does not close spontaneously is not routinely needed. Waiting until this blood vessel, the ductus, closes by itself is just as safe, research coordinated by Radboud university medical center has shown. There are even tentative indications that medication to close the ductus might increase the risk of chronic lung injury.
In preterm infants, the ductus arteriosus, the blood vessel that supplies oxygen to a fetus' body during pregnancy, often does not close spontaneously. Worldwide, these children are commonly treated with the drug ibuprofen after birth so that the vessel closes. For many years, the prevailing thought has been that this prevents complications such as cerebral bleeding or chronic lung injury. However, for a long time uncertainty has existed about the optimal management of a patent ductus in preterm infants. Uncertainty whether the patent ductus should be treated by administering medication or whether a wait-and-see policy is better.
That is why a study, led by Professor of Neonatology Willem de Boode from Radboudumc, started in December 2016. This involved 273 babies born at a gestational age of under 28 weeks. In one group the ductus was closed using ibuprofen, in the other group they waited for the ductus to close by itself. The results have now been published in New England Journal of Medicine, in an article of which PhD student Tim Hundscheid is first author.
The study has shown that expectant management, in which the ductus closes by itself over time, is just as safe as a policy in which treatment is provided. The starting point was the chance that a child survives without serious complications to the lungs or intestines. De Boode: ‘This represents an enormous revolution in our thinking about the ductus. Our dogma is changing.’
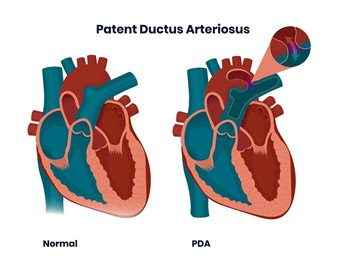
In this image: on the left the heart without open ductus, on the right with patent ductus arteriosus.
Is the cure worse than the disease?
This is not the only result. The researchers found indications that ibuprofen might increase the risk of chronic lung disease. De Boode and colleagues are surprised by this result. ‘We expected to show that an expectant approach would be as good as active closure. But the results tentatively indicate that the use of ibuprofen might even have adverse effects. It seems that the remedy, ibuprofen, might be worse than the condition, the patent ductus.’ The complications we attributed to the open ductus are also known side effects of ibuprofen. ‘To thoroughly understand this, further research is needed’, the neonatologist said.
According to De Boode, we cannot yet conclude that not treating is better than treating. ‘There certainly is a select group of patients that does need treatment. This will be one of the objectives of additional research.’ A possible alternative to medication is transcatheter closure, in which a plug that closes the ductus is inserted through the groin by means of a catheter.
Parental involvement
Willem de Boode stresses the importance of scientific research in preterm infants. ‘We still have a lot to learn about these children. That is why these kind of studies are so important, they increase our knowledge.’ In particular, he is enormously grateful to the parents of children who participated. ‘Most parents were totally overwhelmed by their baby's preterm birth and admission to the Intensive Care. Within the first three days after birth, we asked them if they wanted to participate. I am so very grateful that so many of them wanted to contribute to this research and have given us their trust. Without them, we would never have gained this knowledge.’
About the publication
This article was published in The New England Journal of Medicine: Expectant Management or Early Ibuprofen for Patent Ductus Arteriosus - T. Hundscheid, W. Onland, E.M.W. Kooi, D.C. Vijlbrief, W.B. de Vries, K.P. Dijkman, A.H. van Kaam, E. Villamor, A.A. Kroon, R. Visser, S. M. Mulder-de Tollenaer, B. De Bisschop, P. Dijk, D. Avino, C. Hocq, A. Zecic, M. Meeus, T. de Baat, F. Derriks, T. Brink Henriksen, K. Jacobsen Kyng, R. Donders, D.H.G.M. Nuytemans, B. van Overmeire, A.L. Mulder, and W.P. de Boode for the BeNeDuctus Trial Investigators. DOI:10.1056/NEJMoa2207418.
About the BeNeDuctus trial
The BeNeDuctus trial was conducted within the Neonatology Network Netherlands, the N3 Research Network. This study, which included hospitals from Belgium and Denmark in addition to the nine Dutch centers with a Neonatal Intensive Care Unit, was funded by ZonMw (the Netherlands) and KCE (Belgium). Close cooperation was established with Care4Neo, the association representing the interests of parents whose child is admitted to a hospital immediately after birth.

On this picture: Professor of Neonatology Willem de Boode.
-
Want to know more about these subjects? Click on the buttons below for more news.
More information
Pauline Dekhuijzen

wetenschaps- en persvoorlichter
Related news items

Research sets standard for renal function of newborn babies Abnormal kidney function is now easier to detect
28 April 2022 Researchers at Radboudumc have succeeded in establishing reference values for functioning of the kidneys of newborn babies. This allows for earlier detection of kidney injury and for improved dosing of drugs handled by the kidneys. go to page



