
When tumor cells are stressed by a low level of oxygen in the primary tumor, they go into a kind of power saving mode. In this way, they detach more easily as single cells from the tumor and spread to other parts of the body. Veronika te Boekhorst and Peter Friedl from Radboudumc demonstrate this process and the underlying molecular mechanisms in a publication in Current Biology.
Under normal conditions, breast and head and neck cancer cells search together as multicell masses for sites in surrounding tissue where they can grow and extent the tumor. This process is called collective invasion and is similar to root growth in plants. The team of Peter Friedl, Professor of Microscopic Imaging of the Cell at the Radboudumc, now shows that tumor cells migrate in a different way when there is a lack of oxygen. They then detach from each other, individualize, and leave the tumor to find a better place elsewhere in the body.
"Let’s say we are holding each other’s hands, then we don't move very fast. But when we let each other go, I will move off like a rocket," Friedl explains. "We call this solitary traveling of tumor cells amoeboid movement, because amoebae, organisms that consist of only one cell, also move like this. This is the oldest mode of movement in evolution, and it has been around for more than a billion years. Tumor cells thus resort to an ancient mechanism when they are deprived of oxygen, allowing them to quickly escape the tumor as a single cell."
Fiat Panda
Friedl studied the mechanisms behind this strategy and discovered that the protein calpain plays a crucial role. In a tumor with little oxygen, the amount of calpain goes up. This enzyme regulates the breakdown of so-called integrins. These normally ensure that a tumor cell remains neatly attached to the tumor. The cells therefore become detached. The single cells use little oxygen, up to eighty percent less than normal. In this 'eco-mode' they travel to other parts of the body. After a day or two, cells turn to normal mode again and they grow to form new tumors.
"You can compare it to cars," Friedl explains. "Say you have a Porsche, it uses forty liters of gasoline for a hundred kilometers of distance. But you can also travel in a Fiat Panda. That only uses five liters of gasoline for a hundred kilometers distance. They both get from A to B at the same speed, but the Fiat Panda uses much less energy. This also applies to tumor cells that follow this strategy and break all energy-consuming connections with other cells."
Escape mechanism
In the lab, Friedl observed in a model of breast cancer that normal tumor cells metastasize much less than tumor cells that receive too little oxygen. Metastasis thus appears to be an escape mechanism for the stressed tumor cell. When Friedl added drugs that inhibit the enzyme calpain, the stressed cells started to behave like normal tumor cells again and barely metastasized. Thus, calpain not only puts tumor cells in a state of low oxygen consumption, but also causes them to metastasize.
"Substances that inhibit calpain are therefore very interesting in patients with tumors that are low in oxygen and therefore metabolically stressed," Friedl says. "These drugs are not yet clinically available. Phase 1 studies have yet to start. Hopefully, our findings will accelerate these studies and further research into the possible anti-cancer effect of these compounds."
Live imaging: tumor cells escape from tumor

On the left a normal tumor, where cells seek to expand in a certain direction via collective invasion. On the right a tumor with a lack of oxygen. Cells let go and escape the stressful situation.
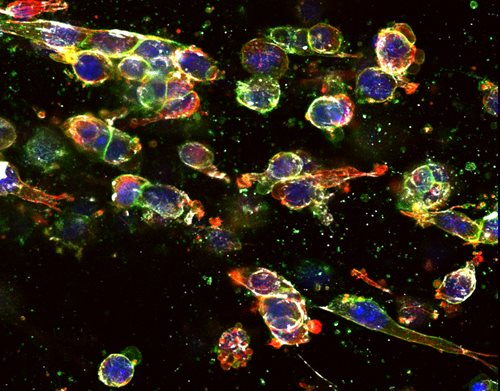
Cells let go of each other and run away. Blue = cell nuclei. Red = actin. White/green = active integrins. When cells are still attached to each other, as in the upper left, we see many more integrins between the cells.
About the Publication
This research was published in Current Biology: Calpain-2 regulates hypoxia/HIF-induced plasticity toward amoeboid cancer cell migration and metastasis. Veronika A.M. te Boekhorst, Liying Jiang, Marius Mahlen, Maaike Meerlo, Gina Dunkel, Franziska C. Durst, Yanjun Yang, Herbert Levine, Boudewijn M.T. Burgering, and Peter Friedl.
-
Want to know more about these subjects? Click on the buttons below for more news.
More information
Annemarie Eek

wetenschapsvoorlichter
Related news items

Willem Mulder appointed professor of Precision Medicine
4 October 2022Willem Mulder has been appointed professor of Precision Medicine at Radboud university medical center / Radboud University. He develops new drugs for immunotherapy against cancer, inflammation, rejection reactions and infectious diseases.
go to page
Immune cell becomes tumor cell by a molecular switch to higher lipid uptake Protein CD37 plays an important role in the development and prognosis of B-cell lymphoma
19 September 2022When B cells of the immune system no longer have the protein CD37 on their surface, they can change into tumor cells that form blood cancer. CD37 inhibits fat uptake in B cells. If this protein is missing, a tumor cell absorbs much more fat and therefore grows faster.
go to page


