RIMLS talent highlights of 2019

STORMy discoveries by our cell biologists

A piece of art by Ben Joosten
Sometimes research, in addition to all the impactful results, simply creates a beautiful piece of art. Ben Joosten made a creative composition with our newest microscopy equipment and won the Best MIC Image award with it. read moreA piece of art by Ben Joosten
This picture shows a human primary dendritic cell labelled for actin cytoskeleton imaged by the home-built super-resolution STORM microscopy compared to the small upper corner detail of the same cell imaged by conventional fluorescence microscopy.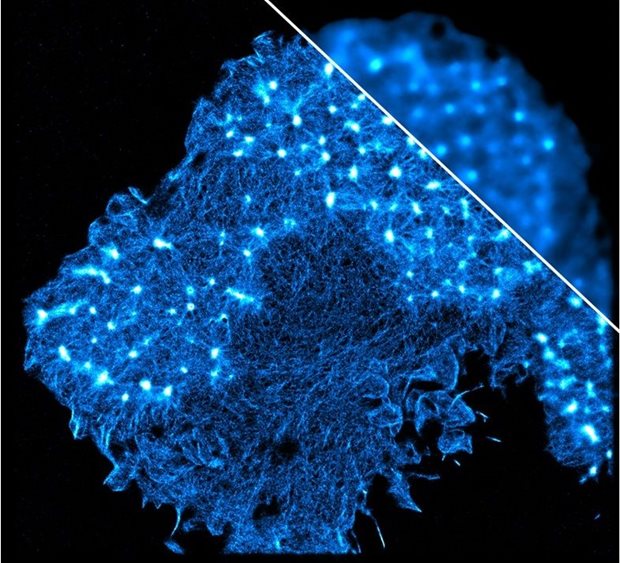
Ben Joosten (Nanomedicine) together with Gert-Jan Bakker (staff scientist high-end microscopy, Cancer development and immune defence) built the STORM setup from scratch. They were able to capture images of intracellular structures and reveal unprecedented details of the cellular organization. In this case, we can appreciate the increased resolution that allows to detect the structural connections between these dot-like cytoskeletal structures called podosomes. Podosomes scan the surface and mediate cell protrusion and migration of dendritic cells.
The STORM setup is now available to all RIMLS scientists and Ben Joosten is the contact person. Considering the incredible efforts made by this remarkable technician who pushed himself outside his own expertise to learn the ins and outs of this complex, state-of-the-art microscopy technique, , Ben Joosten received the well-deserved Best MIC Image award 2020., Ben Joosten received the well-deserved Best MIC Image award 2020., Ben Joosten received the well-deserved Best MIC Image award 2020.

What was the best PhD Thesis in 2019?
The fundamental insights of this thesis by Guido van Mierlo (Faculty of Science) will undoubtedly contribute to a comprehensive understanding of pluripotent cells which will ultimately impact regenerative and personalized medicine. read moreWhat was the best PhD Thesis in 2019?
Guido van Mierlo (Faculty of Science) successfully defended his PhD thesis. The title of his thesis was “Viewing pluripotency from a multi-omics perspective”. His thesis was awarded the prize for the best PhD Thesis of RIMLS in 2019.According to the jury: “The thesis is of excellent quality, coherent, and beautifully illustrated and is composed of a selection of publications in highly renowned journals co-authored by the nominee, one of which has just been submitted, but has been deposited on the open access preprint repository bioRxiv advocating open science. The work describes high quality multidisciplinary science, applying challenging techniques in a “multi-omics” approach to obtain groundbreaking results characterizing the molecular mechanisms of epigenetics and pluripotency. The fundamental insights of this thesis by Guido van Mierlo will undoubtedly contribute to a comprehensive understanding of pluripotent cells and will ultimately impact regenerative and personalized medicine".

Potential for further clinical development?
Julie Verhoef was awarded with the best Master thesis. An excellent thesis which describes a challenging translational project deciphering the molecular mechanism of a new class of anti-malarial drugs. read morePotential for further clinical development?
Julie wrote an extensive report of her MMD master internship at the department of Medical Microbiology, where she has been highly productive and has generated significant scientific data.The jury motivated their choice: “This excellent master thesis is exemplary well outlined and structured, very well readable and excellently illustrated. The work describes a challenging translational project deciphering the molecular mechanism of a new class of anti-malarial drugs. A multidisciplinary approach is described covering cloning, CRISPR/Cas gene editing in a notoriously difficult organism, pharmacology and imaging techniques. The results reported in this thesis contributed to a publication in the journal Science Translational Medicine and shows the potential for further clinical development of a class of anti-malarial agents characterized in the thesis.”

Research with significant scientific impact
This applies to the RIMLS breakthrough paper of 2019, won by Bas van der Sloot and colleagues. The highly innovative method involves the manipulation of antibody genes in the hybridoma cells by the CRISPR/Cas9 genome editing technology in combination with homology-directed repair.
read moreResearch with significant scientific impact
Felix Fenneman, co-author, received the prize on behalf of Bas van der Schoot.
Functional diversification of hybridoma-produced antibodies by CRISPR/HDR genomic engineering. Bas van der Schoot et al., Science Advances 5, 8, aa1822, 2019.
In the RIMLS breakthrough paper of 2019, Bas van der Schoot and coworkers published a novel, generic approach to manipulate the format of antibodies produced by established hybridoma cells. Their highly innovative method involves the manipulation of antibody genes in the hybridoma cells by the CRISPR/Cas9 genome editing technology in combination with homology-directed repair. In this way antibodies can be produced in the format of choice, including chimeric isotype antibodies, mutants of the constant domain, antibodies with affinity tags and antibodies carrying sequence elements for site-directed conjugation. The jury was highly impressed by the wide applicability of this technology, which implicates that this paper will have a significant scientific impact, both in the short and the long term.
The CRISPR/HDR-based platform provides a new toolbox for antibody engineering that appeared to be highly efficient. The method will not only be applicable in scientific research, but will also have important implications for therapeutic antibody development. The site-specifically functionalized antibody molecules will find applications in various fields, such as nanomedicine, cell biology, molecular biology and the generation of antibody-based drugs.
The jury was pleased to see that the authors have published this paper in an open access journal and that the required molecular tools have been made available via Addgene.
Finally, it should be emphasized that Bas van der Schoot and the Verdoes group executed this work in a rather multidisciplinary setting in collaboration with various researchers of other centers in the Netherlands.
