

About
We focus on early detection and treatment of common diseases. Our three focus areas are: anatomy, nuclear medicine and radiology.
Research groups organized by our department
Scientific impact

Ambition and impact
See how our department creates scientific impact that leads to innovation in health and healthcare.
read moreAmbition and impact
We focus on early detection and treatment of common diseases. Our three focus areas are: anatomy, nuclear medicine and radiology.
Our research includes basic research at the molecular level, the development of new medical devices and the development of software tools. We translate these into clinical applications that can be used in daily routine.
-
The Dosimetry Core Unit supports effective and safe radionuclide therapy for cancer patients. Its researchers perform translational research, covering the whole range from bench to bedside.
For more information and services, see their page.

Research programs
Our department is active in several research programs. Use the filter option on the research programs page to see which programs we contribute to.
see page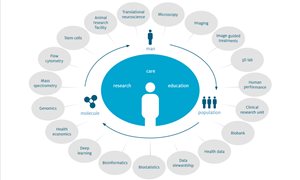
Technology centers to boost your research
See which of the 19 technology centers are affiliated with our department and how they can enhance your research.
read moreTechnology centers to boost your research
The Radboudumc Technology Centers are an access point for technological expertise, high-end equipment and biobanks. They can support a wealth of research questions. The centers are linked to research facilities of both Radboudumc and Radboud University.
See which of the 19 technology centers are affiliated with our department and how they can enhance your research.
-
This technology center provides cutting-edge technology and service for imaging-related preclinical and clinical research questions.
For more information and services, see their page.
Other efforts
Internships
Students of our medical faculty can access OnStage with their RU account. An overview of all internship opportunities is available here.
Are you not a student of our medical faculty? Then you can use our department's general contact information, found at the top of the department page.
Ongoing medical-scientific studies
Organization and people
Getting here
Visiting address
Radboudumc main entrance
Geert Grooteplein Zuid 10
6525 GA Nijmegen
Directions



Vacancies
When research positions become available in our department, they will automatically appear in an overview below.
