About
The goal of orthopedic surgery is to reconstruct deformities and defects of the musculoskeletal system. Our research focuses on the external loads applied to the musculoskeletal system, and how these loads influence the reconstructions.
Aims
The Orthopaedic Research Laboratory (ORL) aims to determine the loads acting on the musculoskeletal system during activities of daily living, and how orthopaedic surgery influences these loads and joint kinematics. For this purpose, computational analyses (e.g. finite element analysis, musculoskeletal modelling) are combined with in vitro testing, and medical imaging ((dynamic) CT and MRI). These tools are used to better predict the outcome of interventions, to further improve the quality of orthopedic care.
To reliably and accurately translate these interventions to the operating room, the ORL works together with MITeC and the 3D Lab to develop surgical guidance systems, navigated surgery, and robotics.
Discoveries
The Orthopaedic Research Lab is a pioneer in the use of finite element modelling (FEM) for bone implants.
read moreDiscoveries
The Orthopaedic Research Lab is a pioneer in the use of finite element modelling (FEM) for bone implants. Its expertise was acknowledged by the European Research Council by ERC-Advanced Grant on Biomechanical Diagnostic, Pre-Planning and Outcome Tools to improve Musculoskeletal Surgery (BioMechTools). The research on this subject is acknowledged worldwide as pioneering and leading research.
Areas of expertise

Experimental testing
We are specialized in mechanical testing, computational analyses, histological analyses and animal testing. Our strength lies in combining these techniques, allowing for cross-validation and innovative research.
read moreExperimental testing
The Orthopaedic Research Laboratory (ORL) performs research in the field of orthopaedics. The biomechanical section focusses on bone, soft tissues and on pre-clinical testing of implants. We are specialized in mechanical testing, computational analyses, histological analyses and animal testing. Our strength lies in combining these techniques, allowing for cross-validation and innovative research.
-
The ORL has one fully calibrated MTS testing machines with loading ranges up to 15 Kn and also has the capability to apply a torque up to 225 Nm. We have capabilities to design dedicated experimental set-ups to assess strength, stability and fatigue properties of various materials and reconstructions. Reconstructions are often tested in combination with cadaveric materials which we obtain from the Anatomical department of the Radboudumc. Testing conditions can vary from dry to simulated body fluids at 37 degrees centrigrade.
-
The ORL has a Bose ElectroForce BioDynamic test machine. This uni-axial loading machine can apply a maximal load of 200 N. Dynamic or static loading can be performed under incubator circumstances. Hence, tissue growth and biological specimens within a sterile environment can be tested, and the biological tissue can be subjected to in-vivo biomechanical loads.
-
We are experts in the applications of radiostereometric analysis (RSA) to quantify the 3-D motion of one segment to the other. This is of high relevance if we want to determine the migration of an implant relative to bone upon dynamic loading, or if we want to determine how much soft tissues deforms due to loading or kinematic changes.
Depending on the experimental conditions the accuracy of this technique is around 50 microns.
-
The ORL utilizes Tekscan pressure mapping to assess how joint loading is altered due to interventions such as cartilage repair, ACL reconstruction, implant positioning and other reconstructive techniques. We have ample experience in calibrating and registering the pressure maps in a reliable way.
-
Digital image correlation and tracking is an optical method that employs tracking and image registration techniques for accurate displacement and strain measurements. We utilize this technique to quantify micro-motions between implant and bones and to determine strains in soft tissues.
-
To measure 3D motions of segments the ORL has the capability of using a Polhemus Fastrak motion tracking system. This system is based on electromagnetic principles which limits the experiments to non-metal investigations. This system can be easily used to quantify joint kinematics and how they change due to an interventions such as placing a prosthetic component or performing a surgical reconstruction. Errors are smaller than 1 mm in translation and 1 degree in rotations.

Computational modeling
Using Computational modeling, we can perform biomechanical analyses of complex orthopaedic problems and interventions. Computational analyses that are routinely used include Finite Element Analysis (FEA) and musculoskeletal modeling (MSM).
read moreComputational modeling
Using Computational modeling, we can perform biomechanical analyses of complex orthopaedic problems and interventions. Computational analyses that are routinely used at the Orthopaedic Research Lab include Finite Element Analysis (FEA) and musculoskeletal modeling (MSM).
-
At the Orthopaedic Research Lab, Finite Element Analysis (FEA) is used to investigate the mechanical consequences of orthopaedic interventions. FEA allows us to investigate how forces are transferred through an implant to the host bone, and how the human body adapts to placement of a total joint replacement. FEA is also used to evaluate the performance of orthopaedic implants, and to judge the strength of bone in case of metastatic lesions.
-
Musculoskeletal modeling is used to simulate the motion of human joints (joint kinematics), and how forces are transferred through the joint (joint kinetics). Musculoskeletal models are composed of rigid bodies representing the human skeleton, actuators representing the muscles, springs representing the joint ligaments, and contact surfaces representing the joints.
Together with Aalborg University and Twente University, the Orthopaedic Research Lab has developed an award-winning musculoskeletal model of the knee joint implanted with a total knee replacement prosthesis.

Translational research
At the Orthopaedic Research Lab, we strive for clinical implementation of the research that is performed. One example is the meniscus replacement implant that was previously developed in a research project coordinated by the ORL, and currently is being clinically evaluated.
Clinical research
Our clinical research focuses on the performance of orthopaedic interventions, such as total joint replacement, scoliosis surgery, orthopaedic oncology, and patellofemoral surgery. We aim to evaluate and improve the outcome of our clinical work, to provide the best possible care for our patients.
Projects
-
Total knee arthroplasty (TKA) is a widely used surgical procedure to restore the knee joint after cartilage disorders as osteoarthritis.
read more
Implant fixation of a PEEK total knee arthroplasty
Total knee arthroplasty (TKA) is a widely used surgical procedure to restore the knee joint after cartilage disorders as osteoarthritis. During this surgical procedure, the damaged cartilage layer is removed and the bones are cut in the correct alignment. The following step is the implantation of the TKA components in the knee. One of the major causes of failure after implantation of the TKA components in the knee is aseptic loosening. Since the fixation of the implant has an important role in this aseptic loosening, it is very important to test this beforehand. Implant fixation can be modelled and tested by doing finite element simulations and cadaveric experiments. During these experiments, the loading profiles of multiple activities will be applied to 3D models of the bone and the implant.
The aim of this project is to investigate the implant fixation of a PEEK total knee arthroplasty. This is done with the development of 3D models of the knee and a PEEK knee prosthesis. To create a proper model, the implant characteristics and patient variability need to be taken into account.
Figure 1. Example of a finite element analysis on the femoral component of a total knee arthroplasty.
Researcher for this project: Corine Post. -
Finite element analysis and cadaveric experiments to investigate the fixation of two types of implants with different interference fit.
read more
The effect of interference fit on the primary fixation of cementless TKA components
Finite element analysis and cadaveric experiments to investigate the fixation of two types of implants with different interference fit.
Total Knee Arthroplasty (TKA) is the most common and successful surgical procedure for a diseased knee joint, offering pain relief and improvement of knee function, and providing a better quality of life for patients. The surgery involves the removal of damaged cartilage and bone, then the TKA components are implanted in the knee. The cementless implants fixation depends on the difference between the internal cuts of the bones and the implant dimensions, which creates an interference fit.
Ideally, a higher interference should provide a better fixation, however, it could also cause bone abrasion and permanent deformation during implantation. Therefore, in this study finite element analysis and cadaveric experiments will be performed to investigate the fixation of two types of implants with different interference fit during two loading conditions by measuring the displacement of the implant relative to the bone (also known as micromotions) at the bone-implant interface.
Researcher for this project: Esther Sanchez Garza. -
Accurate fracture risk prediction is important, because it is difficult for clinicians to differentiate between high and low fracture risk lesions.
read more
Fracture prediction in femoral bone metastases
Accurate fracture risk prediction is important, because it is difficult for clinicians to differentiate between high and low fracture risk lesions.
Patients with advanced cancer often develop bone metastases, and in approximately ten percent, these lesions occur in the femur. Femoral metastases may cause pain and can lead to pathological fractures, which severely affect the quality of life. Local treatment of patients with femoral metastases is based on the expected fracture risk: patients with a low fracture risk are treated conservatively with for example radiotherapy to decrease pain, whereas patients with a high fracture risk are considered for stabilizing surgery to prevent a fracture from occurring. Therefore, accurate fracture risk prediction is important. However, it is difficult for clinicians to differentiate between high and low fracture risk lesions, leading to considerable numbers of under and over treatment. To improve fracture risk prediction, we developed the BOne Strength score, or BOS score; a patient-specific finite element (FE) computer model that calculates bone strength based on patient-specific anatomy and bone quality, obtained from CT scans.
Previously, we demonstrated that the BOS score improved fracture risk prediction in patients, compared to current clinical guidelines. The goal of this project is to initiate clinical implementation of the BOS score by determining whether the BOS score aids in fracture risk prediction and treatment decision making for patients with bone metastases in the femur.
Researcher for this project: Florieke Eggermont. -
Develop and implement a highly patient specific decision aid for patients with hip- or knee osteoarthritis.
read more
Artreuze
Develop and implement a highly patient specific decision aid for patients with hip- or knee osteoarthritis
Hip- and knee osteoarthritis are leading causes of disability resulting in joint pain and stiffness. Total hip- and knee arthroplasty (THA and TKA) are a recommended intervention when conservative management appears to be ineffective and disability is significant. Due to this surgical intervention, quality of life rises while the functionality of the joints is ameliorated extensively. Although THA and TKA are considered safe, complications can interfere with a successful outcome or can even put patients in a life-threatening situation. Study findings show that comorbid diseases and medication use are known to influence complication risk and patient outcome.
Decision aids are commonly used to inform patients and to support the patient and physician to think about the level of experienced pain and discomfort, the personal circumstances and the intended type of surgery. These factors and the complication risk can influence which treatment fits best in the patients individual situation.
Until now, decision aids provide general information about treatment options and complication risk. Since one of the main challenges for physicians is to inform the individual patient by providing personal information about the complication risk, we aim to update the decision aid with a pre-operative risk estimation tool which takes the comorbid diseases and medication use of the individual patient into account.
The aim of this project is to develop and implement a decision aid for patients with hip- or knee osteoarthritis to facilitate shared decision making by providing predicted outcomes which are highly patient specific.
Researcher for this project: Lieke Sweerts. -
Automate the segmentation procedure using deep convolutional neural networks.
read more
Innovative techniques to facilitate clinical implementation of fracture risk prediction of the metastatic femur
Bone is one of the most invaded tissues by the metastases of breast, prostate, lung, kidney, and thyroid cancer. Patients with bone metastases are treated based on fracture risk: patients with a high fracture risk are considered for prophylactic surgery to prevent pathological fracture, whereas patients with low fracture risk are treated with conservative treatment such as radiotherapy for pain management.
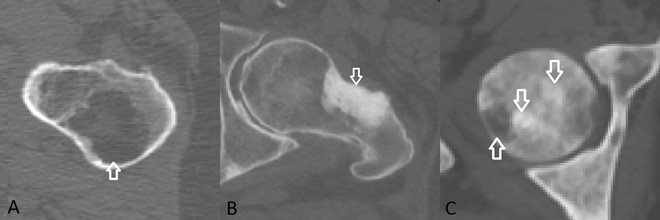 Figure 1: Arrows indicate osteolytic (A), osteoblastic (B), and mixed (C) bone metastases located in the femur.
Figure 1: Arrows indicate osteolytic (A), osteoblastic (B), and mixed (C) bone metastases located in the femur.
Finite element (FE) models can be used to evaluate metastatic bone fracture risk. However, currently, the FE models are not yet applicable to femurs affected with osteoblastic metastases. To determine the mechanical properties within the FE model, calibrated bone densities from CT scans are obtained and since osteoblastic metastases appear very dense on CT scans this results in strong mechanical properties of the metastatic lesions although they are weaker in real life. A potential solution is to assign more appropriate material properties to the osteoblastic lesions to better simulate the mechanical behaviour of the weakened tissue. For this purpose, exact segmentation of the metastatic lesions is important.
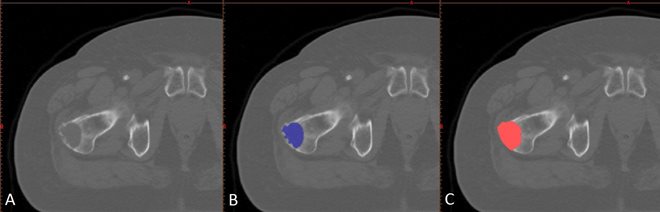 Figure 2: Example of an osteolytic lesion in a right proximal femur (A) and the manual segmentations by two operators (B, C).
Figure 2: Example of an osteolytic lesion in a right proximal femur (A) and the manual segmentations by two operators (B, C).
Manual segmentation is a time-consuming task and it may be difficult to accurately segment every abnormality in the bone structure. The main goal of this study, therefore, is to automate the segmentation procedure using deep convolutional neural networks.
Researcher for this project: Ali Ataei. -
Developing a minimally invasive alternative treatment for patients with metastatic bone disease.
read more
KWF MITEC Project
Patients with metastatic bone disease can suffer from severe pain, pathological fractures of long bones or even spinal cord compression. For these patients, the main focus is on improving their quality of life by providing pain relief and preserving function. At the moment, in order to destroy these lesions, there are two methods available: external beam radiotherapy and open surgery. Radiotherapy doesn’t always succeed in providing pain relief and when it does, it is not immediate.
With the current surgical procedure, pain relief is almost immediate and it eliminates the risk for complications like the ones mentioned above. However, it’s a demanding procedure for a patient who is in a debilitated state. In order to address this, the project focus on developing a minimally invasive alternative treatment. This involves the design of a prototype tool system that can remove the metastatic lesion effectively, reduce risk of tumor reoccurrence and provide structural support to the targeted area. Moreover, it will also include fracture risk predictions and simulations of the impact of the tool to the human bone and surrounding structures for a comprehensive understanding of the tool's performance.
Researcher for this project: Patricia Caetano de Almeida Rodriguez.
Research within MITeC
The Orthopaedic Research Lab has several studies that are partly done within the MITeC environment. For example, the MITeC operating rooms are used for a study on 3D surgical planning and intra-operative navigation for the removal of bone tumors.
read moreResearch within MITeC
The Orthopaedic Research Lab has several studies that are partly done within the MITeC environment. For example, the MITeC operating rooms are used for a study on 3D surgical planning and intra-operative navigation for the removal of bone tumors. This technique will allow better insight in the complete removal of bone tumors in challenging positions, such as the lumbar spine. More complete removal will also result in a smaller change of the tumor recurring.
Another project, focusing on minimal invasive surgical treatment of skeletal metastases, is partly funded by MITeC. The aim is to develop a surgical tool that will allow for the removal, cryotherapy and cementation of skeletal defects. Currently, patients are treated with radiotherapy and analgesics. Thus far, this method shows a partial pain relief with a long onset time. Minimal invasive treatment can possibly improve this.
