20 June 2019
Weak evidence without control group
Radboudumc researchers Machteld van Egmond, Maroeska Rovers, and Niels van Heerbeek are the first to compare the outcomes of two groups of patients following different treatment strategies: septoplasty and non-surgical management, that is medication or watchful waiting. Machteld van Egmond, lead researcher for the project: “This procedure has been performed for many years, but in previous studies only surgical patients were assessed. Without a control group, you never know whether an observed improvement is actually the result of the intervention, or due to other factors, such as the natural course of the condition.”
Two-year follow-up
The researchers investigated the effect of septoplasty in more than 200 adults with nasal obstruction and a deviated septum. The study was performed in two academic medical centers and 16 secondary referral hospitals in the Netherlands. Half of the patients underwent septoplasty (surgical group), and the other half non-surgical management (control group). The researchers then measured the effects of both treatment strategies on patients’ quality of life and airflow through the nose. These measurements were performed at 3, 6, 12, and 24 months after the start of the treatment.
Breathing more easily
The quality of life of the surgical group had already improved at three months. They experienced fewer limitations in daily life due to nasal problems. Nasal airflow also increased after the operation. This positive effect was still present after two years. The patients had fewer nasal symptoms, fewer colds, breathed more easily, and slept better. All the effects were largest at six months after the operation, but persisted until the end of the study.
Publication in Lancet
Septoplasty with or without concurrent turbinate surgery versus non-surgical management for nasal obstruction in adults with a deviated septum: a pragmatic, randomised controlled trial
Machteld M H T van Egmond, Maroeska M Rovers, Gerjon Hannink, Carine T M Hendriks, Niels van Heerbeek
Machteld van Egmond and Niels van Heerbeek are members of theme Reconstructive and regenerative medicine. Maroeska Rovers is member of theme Urological cancers.
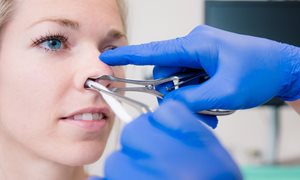 Surgery to straighten a deviated nasal septum, also known as septoplasty, is worthwhile. Patients with a deviated (crooked) septum breathe more easily after this operation and their quality of life improves. The effects of this procedure have never been systematically investigated. Specialists have long debated its benefits. But now, researchers at Radboudumc have ended the controversy on 18 June with a publication in The Lancet.
Surgery to straighten a deviated nasal septum, also known as septoplasty, is worthwhile. Patients with a deviated (crooked) septum breathe more easily after this operation and their quality of life improves. The effects of this procedure have never been systematically investigated. Specialists have long debated its benefits. But now, researchers at Radboudumc have ended the controversy on 18 June with a publication in The Lancet.
Weak evidence without control group
Radboudumc researchers Machteld van Egmond, Maroeska Rovers, and Niels van Heerbeek are the first to compare the outcomes of two groups of patients following different treatment strategies: septoplasty and non-surgical management, that is medication or watchful waiting. Machteld van Egmond, lead researcher for the project: “This procedure has been performed for many years, but in previous studies only surgical patients were assessed. Without a control group, you never know whether an observed improvement is actually the result of the intervention, or due to other factors, such as the natural course of the condition.”
Two-year follow-up
The researchers investigated the effect of septoplasty in more than 200 adults with nasal obstruction and a deviated septum. The study was performed in two academic medical centers and 16 secondary referral hospitals in the Netherlands. Half of the patients underwent septoplasty (surgical group), and the other half non-surgical management (control group). The researchers then measured the effects of both treatment strategies on patients’ quality of life and airflow through the nose. These measurements were performed at 3, 6, 12, and 24 months after the start of the treatment.
Breathing more easily
The quality of life of the surgical group had already improved at three months. They experienced fewer limitations in daily life due to nasal problems. Nasal airflow also increased after the operation. This positive effect was still present after two years. The patients had fewer nasal symptoms, fewer colds, breathed more easily, and slept better. All the effects were largest at six months after the operation, but persisted until the end of the study.
Publication in Lancet
Septoplasty with or without concurrent turbinate surgery versus non-surgical management for nasal obstruction in adults with a deviated septum: a pragmatic, randomised controlled trial
Machteld M H T van Egmond, Maroeska M Rovers, Gerjon Hannink, Carine T M Hendriks, Niels van Heerbeek
Machteld van Egmond and Niels van Heerbeek are members of theme Reconstructive and regenerative medicine. Maroeska Rovers is member of theme Urological cancers.
Related news items
3D-printing prosthesis legs in the middle of the jungle a video call with our colleague Merel in Sierra Leone
14 December 2021 'They sometimes think I'm a sorceress,' says technical physician Merel van der Stelt, while she smiles. 'Take this lady without a lower leg. Yesterday she came here. We scanned her stump and today she already has a 3D printed prosthesis. With a prosthesis people count again'. go to page
Survey of patients to address knee pain from removed meniscus
19 November 2021 Patients with persistent knee pain after meniscus removal can participate at three locations in the Netherlands in the AIR2 study by ATRO Medical, a spin-off of the Radboudumc and DSM. go to page
Turbo Grants for four medical-technical research projects
19 October 2021 Four TURBO grants were recently awarded to new technical-medical research proposals. The grants are part of the TURBO program, a collaboration between the University of Twente (TechMed Centre) and the Radboudumc. go to page
Broken teeth coronastress poses a danger to our teeth
6 May 2021 Teeth grinding and molars on top of each other: stress from corona causes an increase in dental damage. That's what they noticed at the department of dentistry at Radboudumc. go to page
Radboud Science Award for RIHS researchers
24 June 2020 Maroeska Rovers, Machteld van Egmond and Niels van Heerbeek have been awarded the Radboud Science Award 2020. The awardees receive the prize for their research of the necessity of correcting the nasal septum. go to page