
It is the first time that trained immunity is demonstrated after a parasitic infection.
Our immune system consists of two parts; the innate and the acquired immune system. It has long been thought that the acquired part can learn to recognize the pathogens with which it comes into contact, and that the innate part cannot. But in recent years, through the work of Mihai Netea, it has become clear that the innate immune system does have a memory and can therefore be 'trained'.
In this article, trained immunity comes together with another important line of research in the Radboudumc: the infectious disease malaria. From the malaria research Robert Sauerwein, theme Infectious diseases and global health and Henk Stunnenberg, theme Cancer development and immune defence, together with Mihai Netea and other colleagues now demonstrate for the first time that even a parasitic infection can train the immune system. The article has been published in Frontiers in Molecular Biosciences.
The study involved two parts: one part in immune cells, isolated from the blood of healthy volunteers, and a small clinical study in five healthy volunteers infected with malaria parasites (Plasmodium falciparum).
In the blood study, monocytes, immune cells from the innate immune system were isolated. Subsequently, these cells were exposed to malaria-infected red blood cells. After five days the monocytes were stimulated with LPS, a standard way to test trained immunity. It appeared there was a response from the monocytes that had been in contact with the parasite; these cells produce more immuno-active substances such as TNF- α.
The clinical study confirmed these results in healthy subjects. In a controlled way, humans can be exposed to malaria parasites, and the parasites can be examined afterwards. Approximately one month after exposure to malaria, the subjects all showed a similar trained immune response to the response after stimulation with LPS.
About malaria
Malaria, with some 216 million cases and 400,000 deaths per year, is one of the greatest infectious diseases of our time. In recent years there has been an increase in the number of sick people, especially in Sub-Saharan Africa and South America.
The most deadly form of malaria is caused by a single-celled parasite, Plasmodium falciparum, which is transmitted by mosquitoes. Once in humans, the parasite first develops in the liver, for about seven days. The parasite transforms and enters the blood from the liver where it can infect red blood cells. The parasite then develops from an asexual cell into mature male and female reproductive cells. These cells can be sucked up by mosquitoes, after which fertilization of the parasites takes place in the mosquito stomach. The offspring can end up back in humans after a mosquito sting.
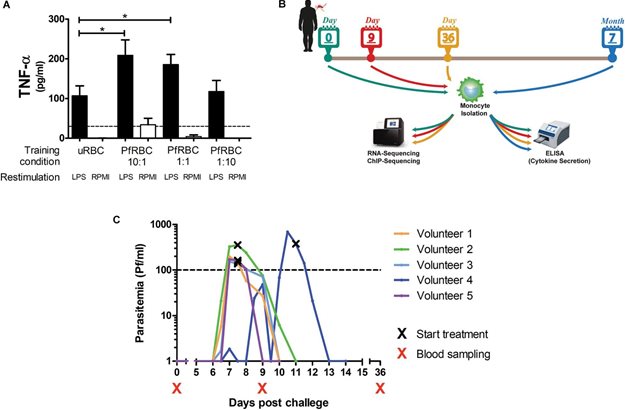
Figure 1. Controlled Human Malaria Infection induces lasting changes in monocytes. Full figure legends can be found in the publication
Related news items

Rubicon grants awarded to three RIMLS researchers
19 April 2022Three researchers have received Rubicon funding from NWO/ZonMw. This will enable Elke Muntjewerff, Laura de Vries and Laurens van de Wiel to do research at a foreign research institute for the next two years.
go to page
RIMLS awards call for nominations
19 October 2021 RIMLS awards several prizes to stimulate and honor our (young) researchers. Upcoming awards are Supervisor of the Year, Best Master Thesis, Best Publication, Best Image and more. Send your nominations now before 24 November 2021. go to page
RIMLS online award ceremony proudly presenting the winners
13 January 2021In this special webinar of the RIMLS New Year Celebration, scientific director René Bindels reviewed 2020 and looked forward to 2021. But more importantly a number of researchers received prizes in the traditional RIMLS awards ceremony.
go to page
Dynamic plasma membrane organization a complex symphony
8 December 2020Sjoerd van Deventer, Abbey Arp and Annemiek van Spriel, published in Trends in cell biology that a dynamic plasma membrane organization is a complex symphony.
go to page
Tumor cells stimulate lipid biosynthesis in macrophages
23 September 2020 RIMLS researchers wrote in the Journal for ImmunoTherapy of Cancer that improved lipid biosynthesis in human tumor-induced macrophages contributes to their protumoral characteristics. go to page